This site is intended for US audiences only.
Evomela®
Stable by design.
Indications and Usage
Evomela® is indicated for use as a high-dose conditioning treatment prior to hematopoietic progenitor (stem) cell transplantation in patients with multiple myeloma.
WARNING: SEVERE BONE MARROW SUPPRESSION, HYPERSENSITIVITY, AND LEUKEMOGENICITY
- Severe bone marrow suppression with resulting infection or bleeding may occur. Controlled trials comparing intravenous (IV) melphalan to oral melphalan have shown more myelosuppression with the IV formulation. Monitor hematologic laboratory parameters.
- Hypersensitivity reactions, including anaphylaxis, have occurred in approximately 2% of patients who received the IV formulation of melphalan. Discontinue treatment with Evomela® for serious hypersensitivity reactions.
- Melphalan produces chromosomal aberrations in vitro and in vivo. Evomela® should be considered potentially leukemogenic in humans.
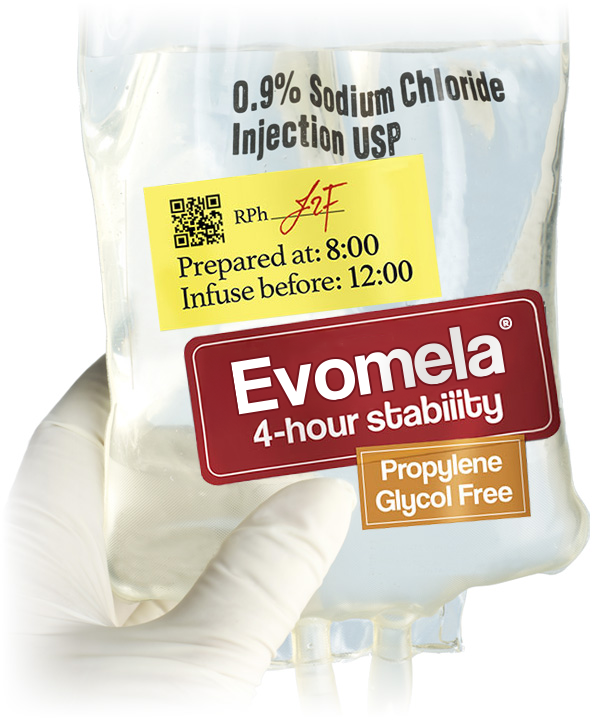
- Evomela® has a propylene glycol–free formulation1-3
- Reconstituted product stable for 24 hours at refrigerated temperature (5°C/41°F), without precipitation1
- Reconstituted product stable for 1 hour at room temperature1
- Admixture solution is stable for 4 hours at room temperature in addition to the 1 hour following reconstitution1
- Administration recommendation: infuse over 30 minutes for conditioning treatment1
References
-
Evomela® Prescribing Information. Acrotech Biopharma Inc.
-
Aljitawi OS, Ganguly S, Abhyankar SH, et al. Phase IIa cross-over study of propylene glycol-free melphalan (LGD-353) and Alkeran in multiple myeloma autologous transplantation. Bone Marrow Transplant. 2014;49(8):1042-1045.
-
Hari P, Aljitawi OS, Arce-Lara C, et al. A phase IIb, multicenter, open-label, safety, and efficacy study of high-dose, propylene glycol-free melphalan hydrochloride for injection (Evomela®) for myeloablative conditioning in multiple myeloma patients undergoing autologous transplantation. Biol Blood Marrow Transplant. 2015;21(12):2100-2105.
- Evomela® was FDA approved based, in part, on its bioequivalence to Alkeran via the 505(b)(2) New Drug Application regulatory pathway1-4
- Evomela® has a PK profile comparable to conventional IV melphalan2
References
-
Evomela® Prescribing Information. Acrotech Biopharma Inc.
-
Aljitawi OS, Ganguly S, Abhyankar SH, et al. Phase IIa cross-over study of propylene glycol-free melphalan (LGD-353) and Alkeran in multiple myeloma autologous transplantation. Bone Marrow Transplant. 2014;49(8):1042-1045.
-
FDA. Center for Drug Evaluation and Research. Evomela® Drug Application. 2016.
-
Hari P, Aljitawi OS, Arce-Lara C, et al. A phase IIb, multicenter, open-label, safety, and efficacy study of high-dose, propylene glycol-free melphalan hydrochloride for injection (Evomela®) for myeloablative conditioning in multiple myeloma patients undergoing autologous transplantation. Biol Blood Marrow Transplant. 2015;21(12):2100-2105.
- Evomela® has a proven safety and efficacy profile for myeloablative conditioning in multiple myeloma patients undergoing ASCT1
- Severe bone marrow suppression with resulting infection or bleeding may occur. Controlled trials comparing intravenous (IV) melphalan to oral melphalan have shown more myelosuppression with the IV formulation. Monitor hematologic laboratory parameters1
- Hypersensitivity reactions, including anaphylaxis, have occurred in approximately 2% of patients who received the IV formulation of melphalan. Discontinue treatment with Evomela® for serious hypersensitivity reactions1
- Melphalan produces chromosomal aberrations in vitro and in vivo. Evomela® should be considered potentially leukemogenic in humans1
Reference
-
Evomela® Prescribing Information. Acrotech Biopharma Inc.



